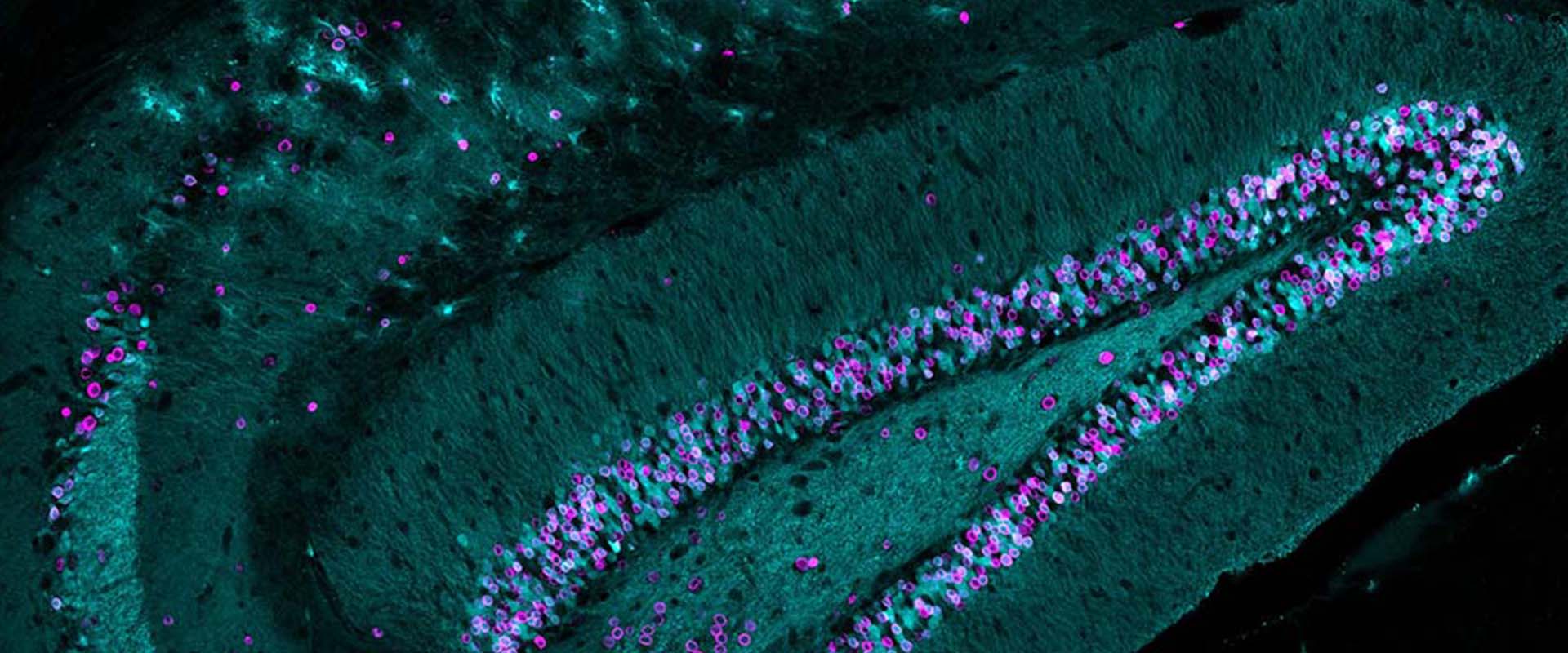
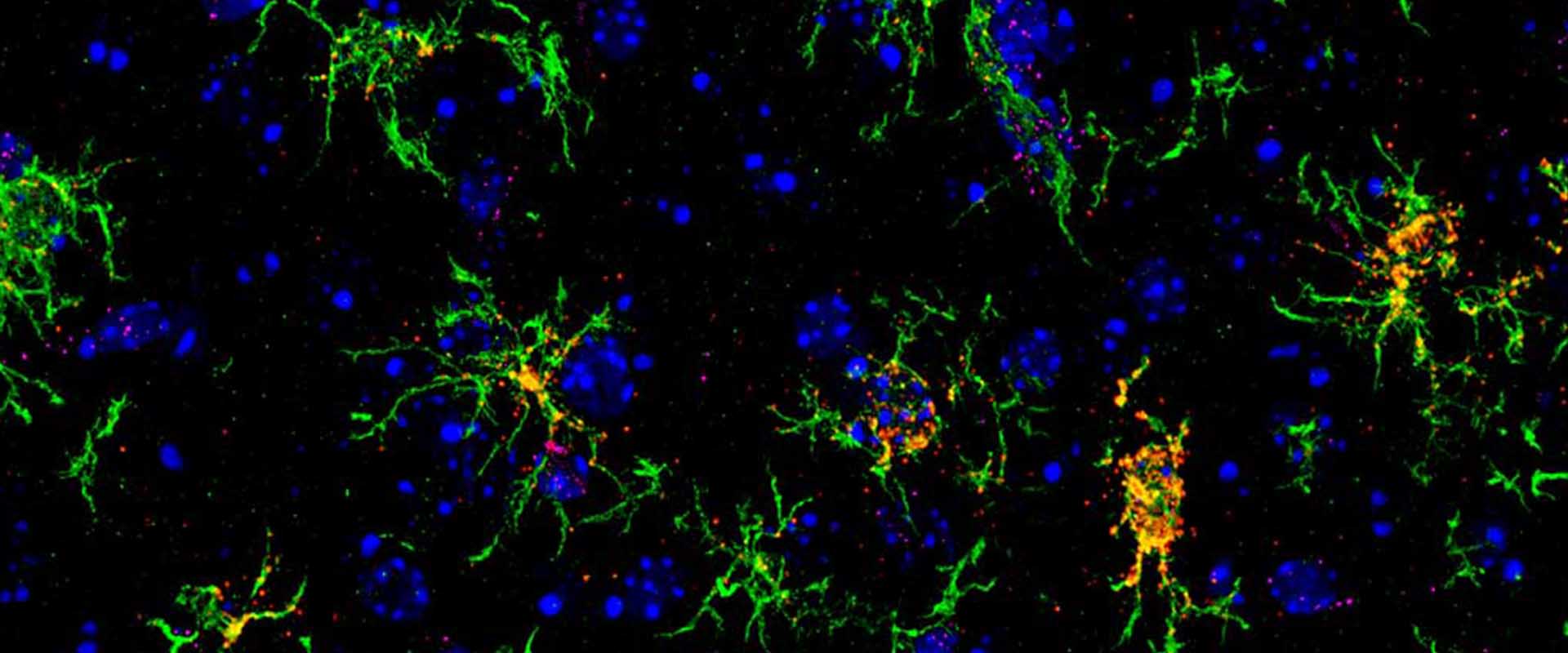
Loss of neuronal 3D chromatin organization causes transcriptional and behavioural deficits related to serotonergic dysfunction
The interior of the cell nucleus is a highly organized 3-dimensional (3D) structure where regions of the genome that are linearly millions of bases apart establishing highly organized sub-structures with specialized functions. Chromatin architecture may have particular importance in neurons because these long-living cells are permanently arrested in interphase and the structures formed during neuronal differentiation are likely irreversible. During neuronal maturation, the nuclear architecture of neural precursors undergoes dramatic changes and passes from a small, heterochromatic nucleus with many small chromocenters to the large euchromatic nucleus with a few large chromocenters characteristic of mature neurons. Although the functional consequences of these changes are obscure, the conserved pattern in terminally differentiated neurons suggests that they may be physiologically relevant. In this study, we generated bi-transgenic mice expressing GFP-tagged histone H2B in principal neurons of the forebrain to investigate neuronal chromatin organization and dynamics in vivo. Surprisingly, the expression of this chimeric histone in mature neurons caused chromocenter declustering and disrupted the association of heterochromatin with the nuclear lamina. The loss of these structures did not affect neuronal viability but was associated with specific transcriptional, electrophysiological and behavioral deficits related to serotonergic dysfunction. Overall, our results demonstrate that the 3D-organization of chromatin within neuronal cells provides an additional level of epigenetic regulation of gene expression that critically impacts neuronal function. This in turn suggests that some loci associated with neuropsychiatric disorders may be particularly sensitive to changes in chromatin architecture.

 English
English